News News
I recently had the opportunity to contribute to a report on one of the most interesting challenges at the intersection of energy and technology: how to power the rapid growth
It was a pleasure to collaborate with EEAE – Greek Atomic Energy Commission to create content tailored to the Greek public regarding radioactive waste and how it is handled in
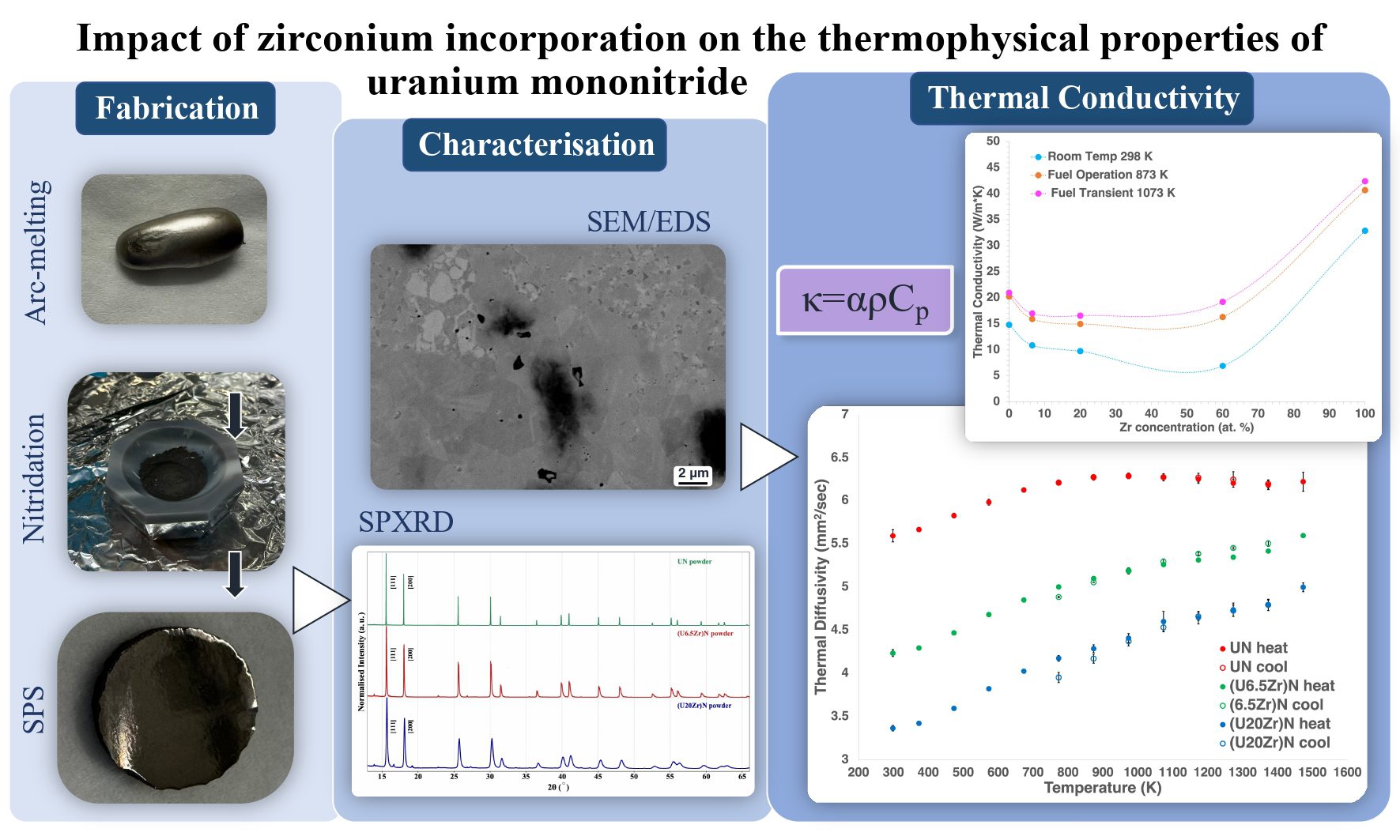
The paper link is here. Elina Charatsidou, Anita Pazzaglia, Kaitlyn Bullock, Maria Giamouridou, Eleanor Lawrence Bright, Mikael Jolkkonen, Christoph Hennig, Pär Olsson Highlights Abstract Uranium mononitride (UN) is a promising candidate fuel for next-generation fast reactors due to
I wrote a chapter in a book that was recently published! Excited to share it with you all! Find the book here:
It was an extraordinary opportunity to present to the council on high-level radioactive waste from North Dakota, speaking both about the SKB KBS-3 method and the importance of nuclear communication
Thank you to WiN Sverige | WiN Sweden for inviting me to their annual conference to host a workshop on gender and science communication. I had an inspiring day at
It was a great pleasure to participate in the Young Generation Sweden meeting in Vattenfall last week. I appreciate the invitation to host a nuclear fuel and science communication workshop
It was a pleasure as always to host a workshop on science communication, this time combined with the gender as peer in the field of nuclear engineering at the NuFuel
It was a great experience to present my most recent work at the NuFuel 2025 conference in Delft, The Netherlands. Exciting works on UN, advanced fuels and great networking opportunities
N. Wikström, M. Giamouridou, E. Charatsidou, P. Olsson, J. Oscarsson, D. Primetzhofer, R.J.W. Frost Abstract: The diffusion of two volatile fission products, xenon (Xe) and krypton (Kr), in zirconia (ZrO) is investigated. Samples of Yttria (YO)-stabilised tetragonal ZrO were implanted with either Xe or Kr, at 300 keV, with a fluence of 1017 at./cm,